KiOmed Pharma obtains CE mark and signs a License and Supply Agreement
KiOmed Pharma announces the granting of the CE mark for its first implantable product intended to treat osteoarthritis symptoms and the signing of a License and Supply Agreement with TRB Chemedica, a leading pharmaceutical and biotechnology company.
KiOmed Pharma announces the granting of the CE mark for its first implantable product intended to treat osteoarthritis symptoms and the signing of a License and Supply Agreement with TRB Chemedica, a leading pharmaceutical and biotechnology company.
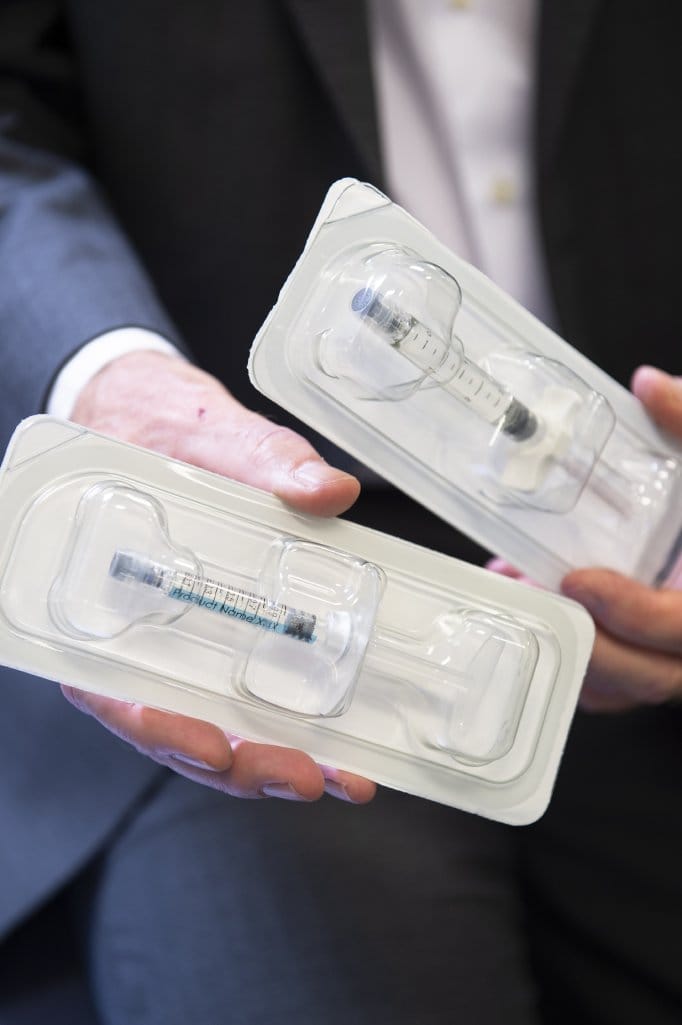
The CE marked product is an innovative intra-articular single injection implant intended to treat osteoarthritic knee pain and other symptoms. It combines unique lubrication of the damaged cartilage and protection of the degraded synovial fluid through the scavenging of free radicals – two main contributors to osteoarthritis pain. Osteoarthritis is a debilitating pathology that affects over 100 million patients worldwide.
Under the signed collaboration agreement, KiOmed grants TRB Chemedica exclusive rights to market and distribute the product across selected markets, and will be entitled to receive upfront, milestones and royalties. TRB Chemedica will also contribute to the clinical and commercial development of the medical device by supporting pre- and post-registration studies and introducing the product in selected territories.
“We are delighted about the achievement of this double objective: CE marking and the signing of this commercial agreement. This will bring our company to the commercial stage and represents a first validation of our vision and ambition to bring a platform of breakthrough products based on KiOmedine® to the market” states François Blondel, Founder and Executive Chairman of KiOmed.
“We are very proud and grateful to our dedicated team and all our partners who have collaborated on this significant development over recent years. We are excited about the partnership with TRB Chemedica and expect this collaboration to result in the treatment and relief of patients awaiting effective treatment of their osteoarthritis pain”
States Houtaï Choumane, CEO of KiOmed

